A huge knowledge base to tap into for information on FDA, HIPAA, SOX, PCI, FISMA, 508 compliance
Designed specifically for the development and implementation of safety and life critical systems
Use as is or use the tools provided to extend, adapt, and elaborate all existing content to make it organization specific
Easy to teach, simple to follow, but productive and practical
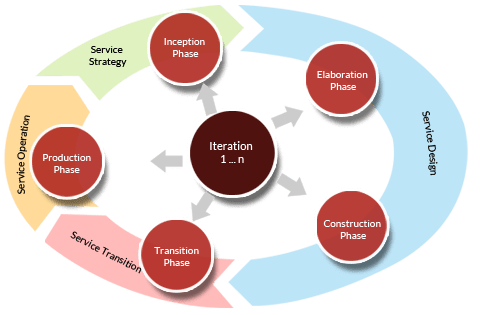
We follow an Agile methodology with time boxed releases designed to be repeatable and easy to document. A well defined lifecycle with simple steps makes it effortless for the team to follow a HIPAA compliant or FDA Quality System Regulated process yet still deliver superior products in a timely manner.

CUP gives you an unfair advantage when meeting FDA, NIST, ONC, or other regulatory mandates
Proving that regulatory mandates are being met in an agile but practical way is not easy. CUP has already gathered all the requirements and put them into a unified approach so that you can immediately start development, not hope for a good Quality System.
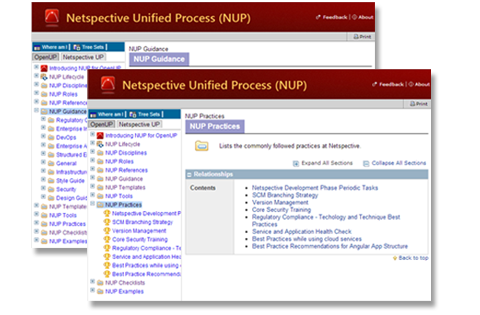
A large body of knowledge is built in, but we give you tools that grow and adapt with your team
CUP is a compilation of practices and guidelines that have been created via significant research and experience. It contains a wealth of knowledge that the team refers to during every phase of a product’s lifecycle. No single process can fit every team so CUP comes with tools that let you extend, adapt, and elaborate all existing content.
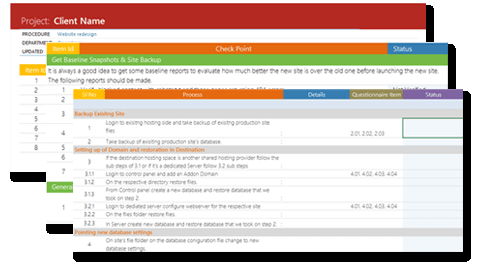
Process verification made easy and fast
Ensures that every process step has been considered.
Ensures that every task has been completed and verifiable.
Ensures that every aspect of quality has been implemented, inspected and the product meets requirements.
Ready to use templates
A huge collection of templates that the team can use to create the artifacts required for every phase of the life cycle.
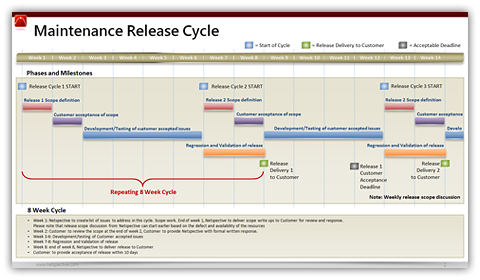
Processes are also organized into disciplines for easy access to resources
Easy to maintain checklists ensure process verification. These checklists are also provided as ready to use checklists in ProveIT.
Roles clearly define the functions of project members.
Well laid out templates for requirements, functional and technical specifications and much more…
A library of references to various resources compiled after extensive research
Processes are also organized into disciplines for easy access to resources
A developer’s guide to tools that can be used in every phase of the lifecycle.
Examples of artifacts created for reference